In order to have a firm grip in the concepts within the broad field of Molecular biology, one has to understand that this study encompasses the attention on those aspects of genetic material and how it is perpetuated and expressed in the forms of RNA and protein. The general topics that will be covered, therefore, are replication, transcription, and RNA processing.
The solid foundation of molecular biology lies in the simple yet complex nature of the DNA structure, which are composed of multiple functional groups and properties, namely:
1) Methyl
2) Hydroxyl
3) Carbonyl
4) Amines
5) Amides
6) Phosphates
Methyls are rather simplistic, its non-polar and it is hydrophobic, it likes water because H20 itself is a polar molecule; and the hydrophobicity of H20 allows the synthesis of hydrogen bonding: so long as there are instances of
a) electronegative atom
b) hydrogen covalently bonded to an electron atom.
Carbonyl's are necessary due to their Carbon to Oxygen double bond, it is polar non-charged but whole group is polar-charged; ionic and Hydrogen bonding interactions are delineated within.
For amines, just know that there are conjugated bases, as well as conjugated acid forms or as what I tell my students (generally known as basic groups).
Amines on the other hand is rather interesting because it is seen in almost every protein structure, both oxygens and nitrogens within in are electronegative, there is a big electron cloud around the whole molecule (or other wise known in biochemical and thermodynamics as "partial" double bound character). Due to the Carbon-Nitrogen partial bond, there is no rotation around this structure, and proteins within our bodies are able to develop a structure due to the C-N bond in the amides.
Phosphates are another key structural component of DNA; it is what makes DNA acidic, it is always attached to a nucleotide, and most phosphates are usually attached to things. One thing that I love to reiterate is the 'Divinity' of phosphates, from it comes all forms of energy: Phosphates are groups where bonding between the phosphate are easily broken AND it is the breaking of these phosphate-phosphate bonds that releases energy -> which in turn allows the making of reactions.
ATP (adenosine triphosphate) are sources of energy that is necessary for every biotic movement is actually a source of energy that is created by phosphates. Without phosphates, proteins will never bond to a DNA sequence of structures, there will be no ribosome organelle that produces the proteins to allow the induction of RNA primers to allow the translation and transcription of the helical bend. That is why we refer to the term "Divinity" of phosphate. Without it, everything in the said organism would be in stasis. Death as a biological and absolute law would manifest in the said organism, species, population, etc.
The Building Blocks: Nucleotides
There are two different types of nitrogenous bases that composes and make up the nucleotides, namely purines and pyrimidine. There are basic structural conformity that differentiates the two, purines are larger 2 ring structures, whereas pyrimidines are a singular aromatic ring.
The 3 major pyrimidines: Cytosine, Thymine => DNA
Cytosine, Uracil => RNA
The 2 major purines: Adenine, Guanine
There are also sugar groups that differs from one another; namely in their grouping
DNA -> Deoxyribose (sugar type)
RNA -> Ribose (sugar type)
Also it is necessary to understand that nucleotides are nucleosides + a phosphates (phosphate + sugar + base)
What is a nucleoside? It is a nucleotide that is missing a phosphate group
The bond between the nitrogenous base and water is called: N-glycosidic bond
When referring to nucleotide know that sugars are labeled in 5' (five prime) to 3' (three prime)
What determines directionality of these compounds?
5'-> 3', it is the sugars
Sugars are joined to each other via a phosphodiester bond, and thus DNA has regions composed of the major grooves and minor grooves. The major grooves are larger spaces observed in the anti-parallel double helix with no sugar or phosphate backbone face it; minor grooves have sugars attached to it.
The following are uploaded images from the text book to better illustrate the said concepts
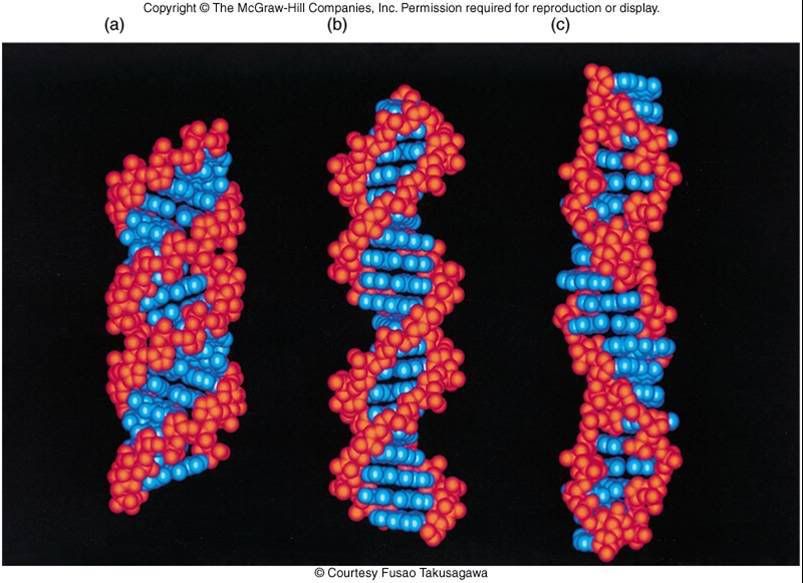
The helical anti-parallel structure of DNA, you can notice the large spaces are the major grooves, the smaller spaces are the minor grooves (which has sugars attached to it)

Proliferation of genetic sequence from a phage parasite onto a host cell; replication explained in here.

The major nucleotidic structures. The top structures, as you can see are 2 aromatic rings, which are part of the purine groups, the lower structures are singular aromatic rings, part of the pyrimidine group. These base groups are what composes the complex sequential nature of our genetic codets. "The fingerprints of GOD," as what I like to say.

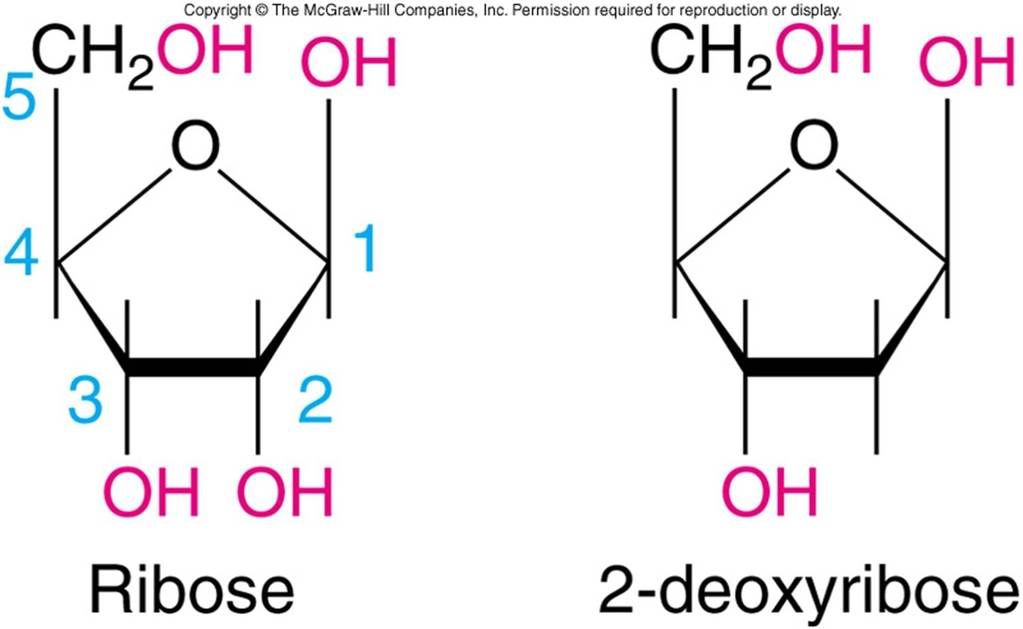
Two sugars. One is the Deoxyribose, the sugar found within eukaryotic species (higher leveled organisms in the echelon of the continuum of life; ex animals, tertiary botanic species)
The other sugar is Ribose, the sugar found within most prokaryotes or 'lesser' organisms, bacteria, virus, fungi etc
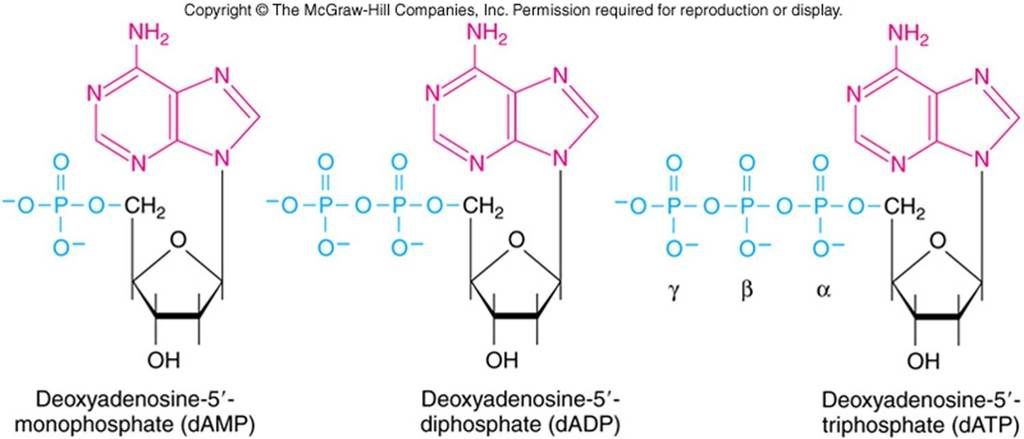
The phosphodiester bonds, the phosphitic bonds within nucleosides. These provide the basis for DNA structure, or energy sequestration, the breakage of these Phosphate bonds literally gives off 'ATP energy' that is essential for life itself. As i said earlier, 'divinity of phosphates'
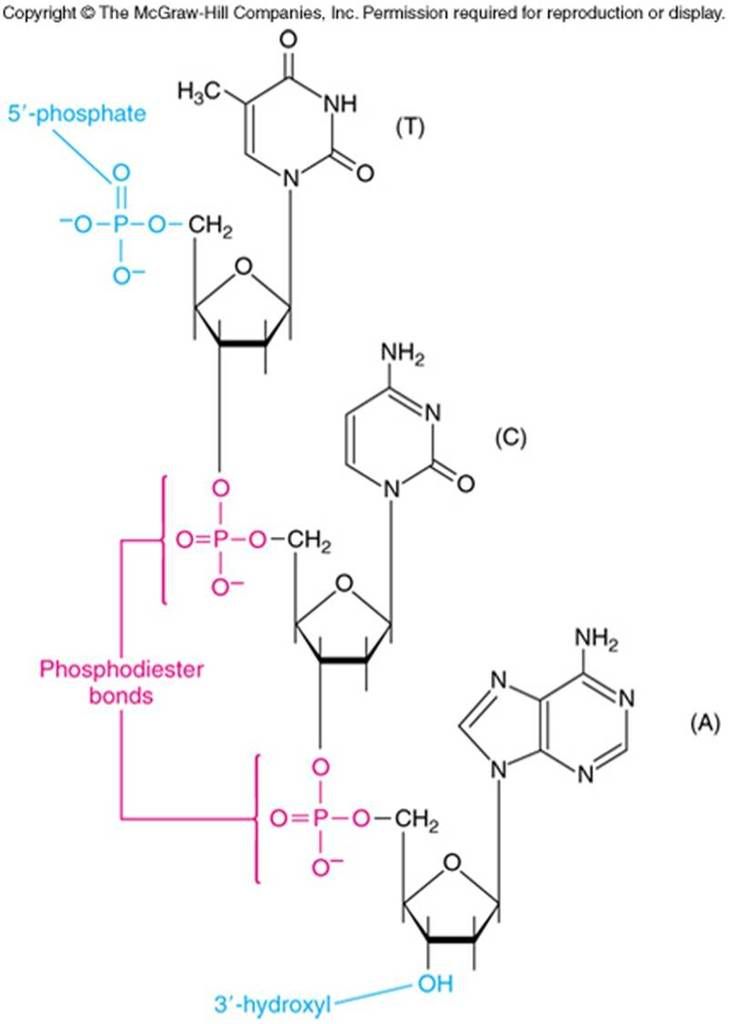
A clearer picture of the phosphodiester bonds
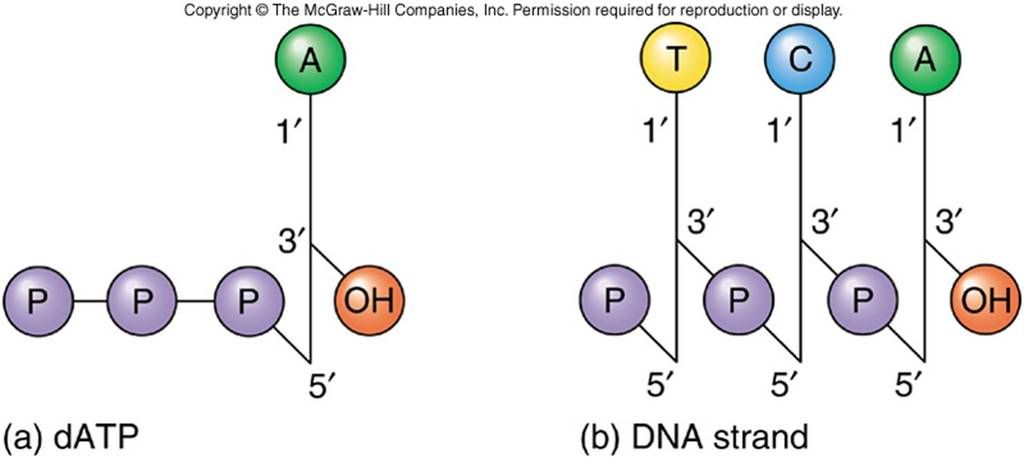
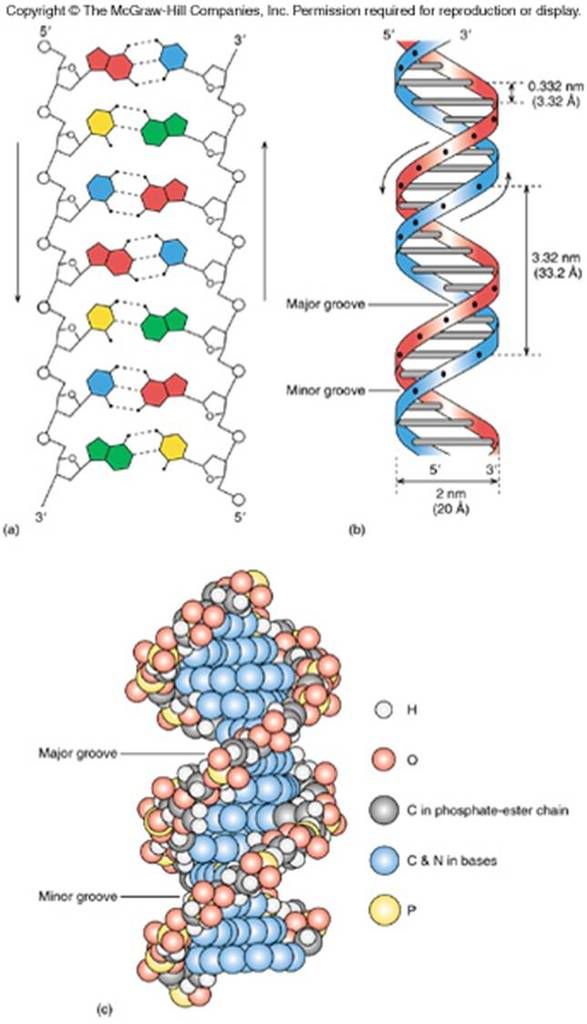
Broader picture, you can actually see in the upper left hand image of the sugars, the phosphates, the nucleotides in full combination, all are complementary. The lower image shows the 'minor' groove, and the 'major' groove. Note that the minor groove has sugars attached to its side, while none are found in the major groove.
Next lesson will be on Protein Structure
Linkback: https://tubagbohol.mikeligalig.com/index.php?topic=8818.0